Johan van Heerden / Assistant professor

Ongoing projects
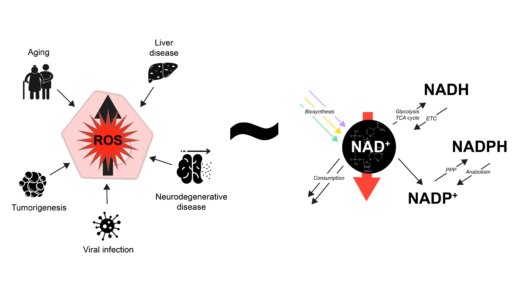
Currently, I mostly divide my time between two research lines. The first aims to understand how NAD+ metabolism is regulated in response to sudden changes in demand for NAD+, an essential cofactor and signalling molecule. Here we employ a combination of computational modelling and experimental work, using hepatocyte-like cell cultures and S. cerevisiae (Baker’s yeast). The second line focuses on growth rate regulation in single bacterial cells, investigating how the fundamental growth laws derived from population-level measurements relate to the growth behaviour of individual cells. To this end, we employ time-lapse microscopy and an in-house-developed generic pipeline for monitoring, segmenting and tracking of individual cells in dense bacterial cultures.
I’m also involved in projects that aim to understand how S. cerevisiae adapts to fast and slow fluctuations in oxygen availability, as well as the development and characterization of genetically encoded fluorescence-based metabolite sensors.
General Research interests
Dynamic (fast) and adaptive (slow) aspects of metabolic regulation
Metabolic and physiological adaptation strategies
Non-genetic origins of phenotypic variability
Coordination of growth and metabolism
Single-cell measurement techniques
Developing and improving experimental methodologies
How is NAD+ metabolism regulated to meet cellular demand?
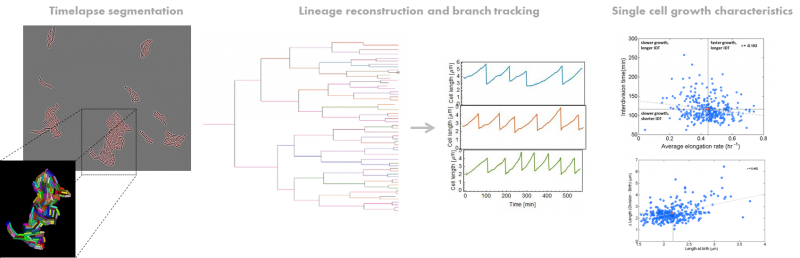
Analysis of single bacterial cells using time-lapse microscopy
To view a summary of the pipeline, click here.
Examples of brightfield time-lapse segmentation for different microorganisms
E. coli
Schizosaccharomyces pombe
Bacillus subtilis
About me
I obtained my B.Sc. (2006) and M.Sc. (2009) degrees in molecular cell biology from the University of Cape Town (South Africa). I started my research career by investigating the potential to exploit gene expression profiles of peripheral tissues as non-invasive diagnostic indicators of psychiatric states, which culminated in my M.Sc. dissertation. In 2009, I joined Prof. Bas Teusink as a PhD student, venturing into the world of microbial physiology and metabolism. In this position, I researched the dynamic regulation of glycolysis in baker’s yeast, focusing on the role that trehalose metabolism plays during transitions from low to high glucose environments.
In 2014, I joined Prof. Frank Bruggeman as a post-doctoral researcher, where I set out to understand how the processes of growth and division impact the stability and variability of phenotypic traits in microbial populations. In this position, I co-developed a pipeline for the automated segmentation and tracking of individual cells in dense bacterial cultures.
Recently, I became a staff member in the Systems Biology Lab, where I am involved in several research projects while continuing to pursue a deeper understanding of metabolism and growth-related processes at the single-cell level.
Contact me
Vrije Universiteit Amsterdam
De Boelelaan 1108, 1081 HZ, Amsterdam
Faculty of Science
Room O|2 01W09
tel: +31 (0)20 598 6993
mail: please click my photo